|
Case Report
An unusual cause of encephalopathy in the setting of a new diagnosis of DLBCL
1 University of Utah School of Medicine, 50 Medical Dr N, Salt Lake City, UT 84132, USA
2 University of Utah Department of Internal Medicine, 50 Medical Dr N, Salt Lake City, UT 84132, USA
3 Medicine in the Division of Hematology at the Huntsman Cancer Center, University of Utah, 2000 Cir of Hope Dr, Salt Lake City, UT 84112, USA
Address correspondence to:
Isabella Frances McNamara
University of Utah School of Medicine, Salt Lake City, Utah 84132,
USA
Message to Corresponding Author
Article ID: 100094Z10IM2021
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
McNamara IF, Rizzi S, Still B, Shah H. An unusual cause of encephalopathy in the setting of a new diagnosis of DLBCL. J Case Rep Images Oncology 2021;7:100094Z10IM2021.ABSTRACT
A 69-year-old woman presented with progressive lethargy and encephalopathy of two weeks duration. Initial laboratory workup revealed hypercalcemia; imaging was notable for a 16 cm by 16 cm right lower quadrant abdominal mass. Core biopsy revealed germinal center Diffuse Large B-Cell Lymphoma (DLBCL). Despite correction of her hypercalcemia, the patient’s encephalopathy persisted. Brain imaging, thyroid stimulating hormone (TSH), folate, B12, urinalysis and culture, antinuclear antibody (ANA), and ammonia were normal. Cerebrospinal fluid (CSF) was negative for leptomeningeal disease. A thiamine level was sent and found to be low. Aggressive repletion was pursued; following the first cycle of chemotherapy, the patient’s mental status normalized. This is a rare case in which the rapid turnover of cells in the setting of active malignancy is believed to play a role in the development of thiamine depletion and resultant encephalopathy.
Keywords: Diffuse Large B-Cell Lymphoma, Encephalopathy, Thiamine
Introduction
Encephalopathy is a broad spectrum of disease secondary to an insult to the brain that changes its function. The differential diagnosis for encephalopathy is broad and can be caused by medications, toxins, metabolites, nutritional deficiencies, and more. Wernicke encephalopathy (WE) and Korsakoff syndrome (KS) are widely recognized complications of excessive alcohol consumption, poor nutritional intake, and resultant thiamine deficiency. However, thiamine deficiency can also be seen in other nutritional deficiencies and high metabolic states such as systemic infection, malignancy, and post-transplant [1]. Thiamine serves as a critical cofactor for many enzymes involved in metabolism. Demand for thiamine positively correlates with high metabolic demand states as well as elevated rates of glucose consumption. Additionally, thiamine deficiency may occur as a result of impaired utilization through inactivating medications, or through excessive losses as reported with diuretic use, diabetes, and hemodialysis [2],[3],[4]. Hematologic and gastric cancers have been described as the most common malignant etiologies of WE/WKS as a result of increased metabolic demand and poor absorption. The classical triad of presentation includes encephalopathy, oculomotor dysfunction, and gait ataxia. However, less than one-third of patients typically present with all three of these findings, making it difficult to recognize [5]. The administration of glucose in these patients exacerbates WE and can lead to the permanent damage of WKS making early recognition of at-risk patients paramount, even in the absence of a definitive diagnosis.
Case Report
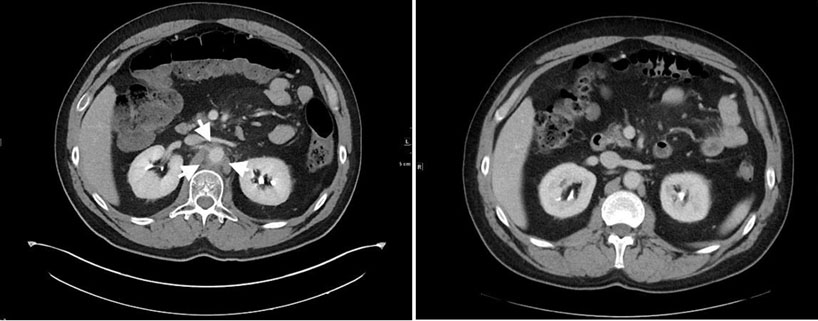
A 69-year-old woman presented with progressive lethargy and encephalopathy of two weeks duration. Initial laboratory evaluation showed a calcium level of 17.2 mg/dL and computed tomography (CT) showed bilateral neck and mediastinal adenopathy, as well as a 16 cm by 16 cm right lower quadrant abdominal mass. A core biopsy of the neck lymph node was performed which showed germinal center Diffuse Large B-Cell Lymphoma (DLBCL). Fluorescent in situ hybridization (FISH) for C-MYC gene rearrangement was negative. Brain magnetic resonance imaging (MRI) and magnetic resonance angiography (MRA) were performed and did not show acute changes or signs of leptomeningeal disease. The patient was treated for hypercalcemia with fluids, calcitonin, and bisphosphonates, then transferred to our facility for persistent encephalopathy and further workup. Her medical history included type 2 diabetes treated with metformin, gastroesophageal reflux disease treated with omeprazole, hyperlipidemia treated with atorvastatin, and hypothyroidism treated with levothyroxine.
The patient was hemodynamically stable on admission. Her electrocardiogram (EKG) and echocardiogram showed normal sinus rhythm and normal heart function. Her hypercalcemia had resolved to 10.8 mg/dL, but her encephalopathy persisted. On exam she demonstrated confusion, was not oriented to time or place, and confabulated. No focal neurological deficits were identified in the cranial nerves, and normal strength was noted throughout her bilateral upper and lower extremities. Her sensation and proprioception were intact, but she demonstrated significant ataxia on ambulation. Labs showed a hemoglobin level of 11.2 g/dL, but white blood cell count and platelets were normal. A metabolic panel showed normal kidney function and liver function tests, but albumin of 3.2 g/dL. Tumor lysis labs and disseminated intravascular coagulation (DIC) labs were normal. Hepatitis B, Hepatitis C, and human immunodeficiency virus (HIV) were negative. All lab values are presented in Table 1.
Workup for encephalopathy, which included TSH, folate, B12, urinalysis and culture, ANA, and ammonia, was unremarkable. An initial lumbar puncture was performed for both CSF analysis and administration of intrathecal methotrexate. Cerebrospinal fluid showed normal cell counts and flow and cytology were negative for malignant cells. Due to the high suspicion for CNS disease, two additional lumbar punctures were performed, which were negative for signs of malignancy. Neurology was consulted and on over-read of MRI brain noted findings concerning for Creutzfeldt-Jakob disease (CJD) given diffuse cortical ribboning, posteriorly dominant bilaterally. Repeat MRI and MRA brain were negative for central nervous system (CNS) disease and did not show concerning signs of CJD. Cerebrospinal fluid meningitis/encephalitis panel, oligoclonal bands, measles, Lyme, treponemal, N-methyl-D-aspartate (NMDA) Ab, voltage-gated K Ab, JC virus, and CSF 14,3,3 all returned negative.
The patient's thiamine level was found to be below the lower limit of normal at 3 nmol/L (normal 4–15 nmol/L), and high dose intravenous (IV) thiamine repletion was initiated at 400 mg q8hr for the first day, then 400 mg daily. Positron emission tomography (PET)/CT showed extensive disease with neck, chest, abdomen, and pelvis with likely right colon involvement. She was initially given prednisone from admission prior to diagnosis. She was then started on R-CHOP with graded introduction to therapy, with D1 Cyclophosphamide, D2 Doxorubicin and Vincristine, and D3 Rituximab. After receiving thiamine replacement and completing the first cycle of R-CHOP with intrathecal methotrexate, her mental status and gait returned to baseline. At her four-month follow-up visit, after 6 cycles of R-CHOP, the patient was found to be in complete remission via PET, and her repeat thiamine level was 223 nmol/L.
Discussion
This is the case of a 69-year-old woman with a new diagnosis of DLBCL who was found to have thiamine deficiency and resultant encephalopathy that we expect were the result of her tumor burden. This clinical presentation of subacute encephalopathy yielded a wide differential diagnosis, including CNS involvement from DLBCL, systemic infection, encephalitis, meningitis, prion disease, delirium, and autoimmune disease. Hypercalcemia was initially presumed to be the underlying etiology given the known correlation between high serum calcium levels and encephalopathy, especially in the setting of hematologic and other malignancies [4]. However, despite correction of her calcium levels, her mental status did not return to baseline. The most notable finding during her workup was a low thiamine level. We did not initially suspect thiamine deficiency or Wernicke’s encephalopathy (WE) given her lack of risk factors including lack of alcohol use, however we did later learn that her overall oral intake had decreased significantly in the two-week period leading up to hospitalization. A number of the medications that she was on at the time of hospitalization including metformin, omeprazole, and levothyroxine have been shown to have loose associations with thiamine deficiency [6]. In the setting of a lack of suspicion for WE, the patient was unfortunately given glucose prior to thiamine replacement. Notably, the patient’s mental status improved modestly after the initiation of thiamine replacement, and ultimately returned to baseline at the time of her follow-up visits during chemotherapy. Additionally, we noticed early significant improvement in her gait following the initiation of thiamine replacement. This improvement in neurological status, along with the corroborating evidence of a lack of thiamine-rich foods in her diet for the period of time leading up to her presentation, as well as a lack of other identifiable causes, led us to believe that her thiamine deficiency was the cause of her altered mental status.
A number of reports have detailed the development of WE or WKS in the setting of treatment of solid and hematological malignancies, however, few have described it as a possible presentation of illness. Wernicke’s encephalopathy has been previously demonstrated in patients with DLBCL [7]. Turner et al. [7] describe a case in which a 37-year-old female with a new diagnosis of DLBCL was found obtunded with ophthalmoplegia and gait disturbances and diagnosed with WE following her 3rd cycle of R-CHOP therapy. As in our report, this patient did not have a recent history of alcohol use, but they did have ongoing active malignancy and dietary changes significant for a decreased intake of thiamine-rich foods for a number of months prior to diagnosis. The patient was treated with intravenous and then oral thiamine, with rapid resolution of her ophthalmoplegia and improvement in gait.
Conclusion
Our case adds to the growing body of evidence that clinicians should be aware that WE/WKS may occur in any patient with poor nutrition and high metabolic demand, especially in the setting of dextrose administration. Our patient presented with three out of four of the Caine diagnostic criteria for Wernicke encephalopathy, each of which returned to its baseline after aggressive thiamine repletion was pursued. Moreover, the treatment of the underlying cancer will reduce the catabolic demand placed on the patient and aid in recovery. We recommend keeping thiamine-deficient states such as WE/WKS on the differential in encephalopathic patients with malignancies and other highly catabolic states. Additionally, patients should aim to include thiamine rich foods in their diet which include grains, meat, and fish.
REFERENCES
1.
Isenberg-Grzeda E, Kutner HE, Nicolson SE. Wernicke-Korsakoff-syndrome: Under-recognized and undertreated. Psychosomatics 2012;53(6):507–16. [CrossRef]
[Pubmed]

2.
Isenberg-Grzeda E, Alici Y, Hatzoglou V, Nelson C, Breitbart W. Nonalcoholic thiamine-related encephalopathy (Wernicke-Korsakoff Syndrome) among inpatients with cancer: A series of 18 cases. Psychosomatics 2016;57(1):71–81. [CrossRef]
[Pubmed]

3.
Zuccoli G, Pipitone N, Santa Cruz D. Metronidazole-induced and Wernicke encephalopathy: Two different entities sharing the same metabolic pathway? AJNR Am J Neuroradiol 2008;29(9):E84. [CrossRef]
[Pubmed]

4.
Brown G. Defects of thiamine transport and metabolism. J Inherit Metab Dis 2014;37(4):577–85. [CrossRef]
[Pubmed]

5.
Caine D, Halliday GM, Kril JJ, Harper CG. Operational criteria for the classification of chronic alcoholics: Identification of Wernicke’s encephalopathy. J Neurol Neurosurg Psychiatry 1997;62(1):51–60. [CrossRef]
[Pubmed]

6.
van Zaanen HC, van der Lelie J. Thiamine deficiency in hematologic malignant tumors. Cancer 1992;69(7):1710–3. [CrossRef]
[Pubmed]

7.
Turner JE, Alley JG, Sharpless NE. Medical problems in patients with malignancy: Case 2. Wernicke’s encephalopathy: An unusual acute neurologic complication of lymphoma and its therapy. J Clin Onc 2004;22(19):4020–2. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Acknowledgments
Isabella F. McNamara was the primary author of this paper, with Scott A. Rizzi, MD and Bryan Still, MD also contributing significantly to both adding content and editing. Harsh Shah, DO oversaw the writing of this manuscript and provided critical feedback.
Author ContributionsIsabella Frances McNamara - Conception of the work, Design of the work, Acquisition of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Scott Rizzi - Conception of the work, Design of the work, Acquisition of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Bryan Still - Conception of the work, Design of the work, Acquisition of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Harsh Shah - Conception of the work, Design of the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2021 Isabella Frances McNamara et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.