|
Case Report
A multifaceted presentation of disseminated coccidioidomycosis: A case review
1 High School Student, Vista Del Lago High School, 1970 Broadstone Pkwy, Folsom, CA 95630, USA
2 Associate Professor, Department of Radiology, UC Davis Medical Center, 4860 Y Street #3100, Sacramento, CA 95817, USA
3 Assistant Professor, Department of Pathology, UC Davis Medical Center, 4400 V Street, Sacramento, CA 95817, USA
Address correspondence to:
Priti Soin
MD, Assistant Professor, Department of Pathology, UC Davis Medical Center, 4400 V Street, Sacramento, CA 95817,
USA
Message to Corresponding Author
Article ID: 100097Z11SK2026
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Kochar S, Ghasemiesfe A, Soin P. A multifaceted presentation of disseminated coccidioidomycosis: A case review. J Case Rep Images Pathol 2026;12(1):1–5.ABSTRACT
Introduction: Coccidioidomycosis, commonly known as Valley fever, is an infectious disease caused by two highly virulent fungi, Coccidioides immitis and C. posadasii. Around 60% of the infections are asymptomatic, however one-third of the cases develop pulmonary infection. About 0.5–2% of the cases disseminate to extrapulmonary locations which can be skin, bones/joints, and central nervous system, frequently seen in immunocompromised individuals (e.g., transplant recipients, HIV patients, pregnancy, diabetics, and patients on steroids).
Case Report: We report a case of coccidioidomycosis in a 33-year-old diabetic male who presented with extrapulmonary manifestations involving thigh and facial skin followed by right chest wall complex collection which disseminated further to rib bone. The patient was found to be non-compliant on his antifungal drug regime which ultimately led to dissemination of the infection in the setting of preexisting diabetes. Following a surgical debridement procedure of the chest wall lesion and rib excision in addition with compliant antifungal regime led to eradication of the infection.
Conclusion: The diagnosis of coccidioidomycosis relies upon clinical suspicion, laboratory findings (serology, cultures, and histopathology) coupled with imaging findings. The key to eradicating infection is ultimately based on an appropriate course of antifungal antibiotics and patient compliance following an accurate and timely recognition of the infection.
Keywords: Coccidioidomycosis, Extrapulmonary, Virulent fungi
Introduction
Coccidioidomycosis is an invasive infectious disease caused by fungi within Ascomycetes division, namely Coccidioides immitis and C. posadasii [1]. The disease was first recognized by Alejandro Posadas, an intern in Buenos Aires, Argentina [2]. The terms Coccidioides (resembling Coccidia) and immitis (not mild) were coined by Casper Gilcrest and Emmet Rixford [3]. Epidemiological evaluations over the years provided significant data on disease’s incidence, risk factors, transmission modes, incubation period, and clinical symptoms [4],[5].
In 2002, a second species, C. posadasii was recognized and named after the original describer of the disease [6]. Both the species are morphologically similar and share 90% homology with respect to the proteins, along with similarities in phenotypes, clinical presentations, in vitro susceptibility, and response to antifungal treatments. C. immitis exists in California, Utah and Washington state, whereas C. posadasii is found in Nevada, Arizona, New Mexico, West Texas, Mexico, and Central and south America [7],[8]. Thus, to date, the differentiation of the two species carries more of an epidemiological and research significance and does not exhibit differences in terms of clinical presentations, in vitro susceptibilities, or response to antifungal treatment.
Case Report
A 33-year-old male who had a prior history of type 2 Diabetes Mellitus for past eight years was diagnosed noticed an enlarging lesion on his left leg which ruptured and spontaneously healed. This was followed by sudden onset of fatigue, weight loss, and night sweats. There were no respiratory symptoms at this point. He also started complaining of pain in the right hip at that time. His treatment history at this point was unknown. Two months later, he presented with non-healing facial wound which was neither responsive to incision and drainage nor several courses of antibiotics. His blood glucose levels at this point were 214 mg/dL (fasting) and his HbA1c was 7.3.
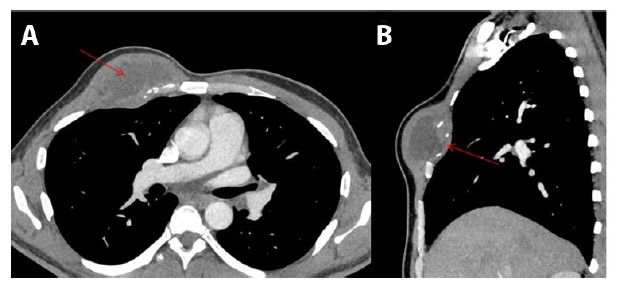
The facial symptoms worsened over a span of next two days for which he presented to the emergency department with the non-healing facial wound and cellulitis accompanied by fever and myalgia. Computed tomography (CT) scan revealed an anterior inferior right facial cellulitis along with bilateral cervical lymphadenopathy (images not shown). Wound cultures were performed which came back positive for Coccidioides (serum antibody titers of 1:4096). The patient was treated with Bactrim and Fluconazole; the latter was given for about five months; however, the symptoms did not improve. Subsequently, he complained of a fluctuating lesion in his right leg, accompanied by hip pain. Computed tomography scan revealed a small abscess with a communication to the skin within the subcutaneous fat of the right lateral thigh, bony erosion of the left parasymphyseal pubic body, right greater trochanter, and enlarged right pelvic sidewall low density necrotic lymph nodes. With the ongoing symptomatology and refractoriness to Fluconazole, Posaconazole was added to the antifungal regime. Subsequently, the patient developed anterior right chest wall complex collection measuring 6.4 cm and involving the right pectoralis muscle with extension into the third anterior intercostal extra pleural space (Figure 1A and Figure 1B). Despite the ongoing antibiotic treatment and supportive measures, the infection spread to the parietal pleura. History of significant weight loss (about 50 lb) within a span of few months was present as well. Additionally, patient’s blood glucose levels were variable and ranged from 113 to 353 mg/dl during the course.
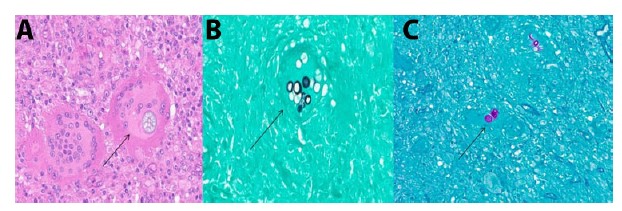
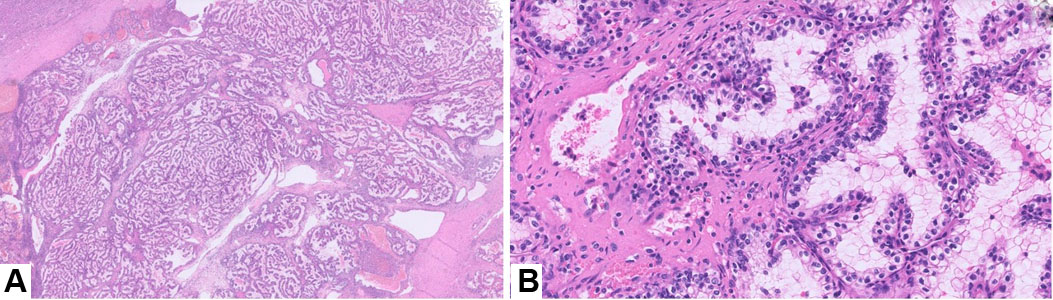
Posaconazole levels were performed to assess patient compliance and surprisingly the levels were subtherapeutic (<0.01) despite an 8-month long course of treatment of Posaconazole. This brought up concerns for patient’s non-compliance for the therapy. The patient’s deteriorating condition necessitated surgical intervention in form of complete debridement of the right chest wall, excision of the right anterior third rib and chest wall reconstruction. During this time, amphotericin was given for a two-week period. Surgical pathology of the rib specimen demonstrated fungal organisms consistent with coccidioidomycosis (Figure 2A, Figure 2B, Figure 2C) along with features of acute on chronic osteomyelitis. The patient was later counseled about the significance of drug compliance following which he drastically improved.
Discussion
Coccidioides spp. are classified as dimorphic fungi which exist in a mycelium form (in soil) and spherule form (in human body). Alternating hyphal form undergoes autolysis, which results in barrel shaped arthroconidia. Inside the human body, arthroconidia transforms to spherules which is the predominant form of dimorphic fungus. Multiplication occurs in the spherules to form 100–300 endospores which are released on the rupture of the spherule [9],[10].
The infection primarily occurs in the lung via inhalation of airborne arthroconidia. Person to person transmission does not occur. Other uncommon modes of transmission include cutaneous inoculation, through organ transplantation and neonatal transmission [11],[12],[13]. Though significant number of infections are asymptomatic (~60%), people with low immunity (e.g., transplant recipients, HIV, pregnancy, diabetics, and patients on steroids) are at risk for disseminated infections. Innate and adaptive immune responses play a vital role in disease severity and dissemination. First and foremost, the initial immune responses to the fungus involve neutrophils, macrophages, and dendritic cells. Additionally, cellular immunity [Th (T-helper) cells and Th17] is critical for containment of infection within the lungs and prevention of dissemination. Thus, immunocompromised population is highly susceptible for Disseminated Coccidioidomycosis [1]. In our case, the patient was a known diabetic with fluctuation levels of blood glucose, posing a significant factor in dissemination of the infection.
Clinical presentation: Clinical manifestations among the symptomatic patients include respiratory illness with typical incubation period of 1–3 weeks. It is a leading cause of community acquired pneumonia in highly endemic population. Symptoms of coccidial pneumonia include cough, chest pain, dyspnea and fever, weight loss and fatigue [1]. Desert rheumatism refers to triad of fever, arthralgia and erythema nodosum. The arthralgias are typically symmetrical and involve lower extremities. The triad is common in women and is usually associated with good prognosis [4]. Immunosuppressed individuals may sometimes present with complicated respiratory manifestations ranging from pulmonary infiltrates, fibrocavitory disease, hydropneumothorax, and respiratory failure [1].
Disseminated disease is involvement outside the pulmonary system and pleural space. The common sites affected by disseminated disease include skin, bones, joints, and central nervous system. Skin manifestations are usually chronic non healing lesions or abscess at any site. Osseous dissemination accounts for about 10–50% of cases of disseminated coccidioidomycosis and is usually associated with presence of systemic diseases as diabetes. This often poses as a diagnostic and therapeutic challenge. Surgical debridement along with appropriate antifungal treatment is usually required for complete resolution [14]. Joints involvement typically occurs in knee and ankles and present with effusion and synovitis. Lumbar vertebral disease is a common location for disseminated disease. Central nervous system (CNS) infection is the most serious form of disseminated disease and can manifest as headache, confusion, gait disturbances, change in mental status, focal neurologic damage or rarely stroke [1]. In our case, the infection involved the facial skin presenting as a refractory abscess which later disseminated to involve thigh skin, multiple bones (parasymphyseal pubic body, right greater trochanter) followed by involvement of the right chest wall and rib. Disseminated coccidioidomycosis can present with multisystem involvement with variable presentations (Table 1).
Diagnosis: Accurate and timely diagnosis of coccidioidomycosis is vital to avoid unnecessary treatment with multiple antibiotics. Effective diagnosis rests upon strong clinical suspicion based on clinical history and symptoms, imaging followed by lab investigations including serology and microbiological cultures and histopathology, the latter two being modes of definitive diagnosis.
Basic laboratory parameters which are usually deranged as in any infection include elevated C-reactive protein (CRP), erythrocyte sedimentation rate (ESR), and hyperglobulinemia. White blood cell (WBC) count is usually variable [1]. Rarely hypercalcemia can be seen [19]. Widely used serological tests are based on detection of anticoccidiodial antibodies.
These are the various confirmatory tests for detection of the infection:
- Detection of Coccidioides-specific nucleic acid in a clinical specimen using a validated molecular assay [e.g., polymerase chain reaction (PCR), deoxyribonucleic acid (DNA) Probe], OR
- Detection of Coccidioides-specific proteins in a clinical specimen or isolate using a validated molecular assay [e.g., matrix-assisted laser desorption ionization-time of flight (MALDITOF)], OR
- Detection of coccidioidal antibodies in serum, cerebrospinal fluid (CSF), or other body fluids using any of the following diagnostic tests:
- Immunodiffusion (ID)
- Complement fixation (CF) with a titter of >1:2
- Tube precipitin
- Detection of both immunoglobulin M (IgM) and immunoglobulin G (IgG) by enzyme immunoassay (may be abbreviated as EIA or ELISA) [20].
Cultures can be obtained from various body fluids like sputum, bronchoscopy fluid, pleural fluid, synovial fluid, CSF, blood, or infected tissues. The colonies grow within a week and demonstrate a white to gray cottony aerial mycelium on agar plate. Barrel shaped arthroconidia can be identified with lactophenol cotton blue preparation [20].
Tissue diagnosis by histopathology almost in all cases shows granulomatous inflammation with multinucleated giant cells with or without necrosis. Dense plasma cells and lymphocytic infiltrates are present. However, these findings can be absent in immunocompromised people [21]. Coccidioidomycosis is identified by spherules filled with endospores on H&E stain. Special stains include GMS and PAS which highlight the above findings and are confirmatory [20].
Conclusion
Coccidioidomycosis is a dimorphic and invasive fungal infection found in endemic belts of Southwestern USA particularly Arizona and Central and southern California. Though a large proportion of the infected population remains asymptomatic or present with mild respiratory symptoms, the disease can take a progressive course in immunocompromised population.
Such disseminated infections in immunocompromised people can have varied multiorgan involvement including osseous involvement and can further have complications and long term sequalae. Accurate and timely diagnosis based on clinical presentation in adjunct with radiology and pathology findings serves as an effective tool toward disease detection. Additionally, treatment with effective antifungal antibiotic and supportive treatments which must be couple with patient compliance is the key for eradication of the infection.
REFERENCES
1.
Crum NF. Coccidioidomycosis: A contemporary review. Infect Dis Ther 2022;11(2):713–42. [CrossRef]
[Pubmed]

3.
4.
Smith CE, Beard RR. Varieties of coccidioidal infection in relation to the epidemiology and control of the diseases. Am J Public Health Nations Health 1946;36(12):1394–402. [CrossRef]
[Pubmed]

5.
Hirschmann JV. Charles Edward Smith: Coccidioidomycologist and public health leader. J Med Biogr 2020;28(1):24–30. [CrossRef]
[Pubmed]

6.
Fisher MC, Koenig GL, White TJ, Taylor JW. Molecular and phenotypic description of Coccidioides posadasii sp. nov., previously recognized as the non-California population of Coccidioides immitis. Mycologia 2002;94(1):73–84.
[Pubmed]

7.
Sharpton TJ, Stajich JE, Rounsley SD, Gardner MJ, Wortman JR, Jordar VS, et al. Comparative genomic analyses of the human fungal pathogens Coccidioides and their relatives. Genome Res 2009;19(10):1722–31. [CrossRef]
[Pubmed]

8.
Hamm PS, Hutchison MI, Leonard P, Melman S, Natvig DO. First analysis of human Coccidioides isolates from New Mexico and the southwest Four Corners region: Implications for the distributions of C. posadasii and C. immitis and human groups at risk. J Fungi (Basel) 2019;5(3):74. [CrossRef]
[Pubmed]

9.
Chow NA, Kangiser D, Gade L, McCotter OZ, Hurst S, Salamone A, et al. Factors influencing distribution of coccidioides immitis in soil, Washington State 2016. mSphere 2021;6(6):e0059821. [CrossRef]
[Pubmed]

10.
Kirkland TN, Fierer J. Coccidioides immitis and posadasii; A review of their biology, genomics, pathogenesis, and host immunity. Virulence 2018;9(1):1426–435. [CrossRef]
[Pubmed]

11.
Wilson JW, Smith CE, Plunkett OA. Primary cutaneous coccidioidomycosis; The criteria for diagnosis and a report of a case. Calif Med 1953;79(3):233–9.
[Pubmed]

12.
Wright PW, Pappagianis D, Wilson M, Louro A, Moser SA, Komatsu K, et al. Donor-related coccidioidomycosis in organ transplant recipients. Clin Infect Dis 2003;37(9):1265–9. [CrossRef]
[Pubmed]

13.
Charlton V, Ramsdell K, Sehring S. Intrauterine transmission of coccidioidomycosis. Pediatr Infect Dis J 1999;18(6):561–3. [CrossRef]
[Pubmed]

14.
Tsantes AG, Koutserimpas C, Naoum S, Drosopoulou LP, Papadogeorgou E, Petrakis V, et al. Diagnosis, treatment, and outcome of coccidioidal osseous infections: A systematic review. J Fungi (Basel) 2024;10(4):270. [CrossRef]
[Pubmed]

15.
Ticknor I, Cadavona JJ, Roby KD, Cotter DG. Disseminated cutaneous coccidioidomycosis masquerading as acne keloidalis nuchae. JAAD Case Rep 2023;39:64–66. [CrossRef]
[Pubmed]

16.
Dhital S, Raza N, Heidari A. A case of disseminated coccidioidomycosis: When the treasure chest wall opened. J Investig Med High Impact Case Rep 2021;9:23247096211040629. [CrossRef]
[Pubmed]

17.
Ramanathan D, Sahasrabudhe N, Kim E. Disseminated coccidioidomycosis to the spine—Case series and review of literature. Brain Sci 2019;9(7):160. [CrossRef]
[Pubmed]

18.
Nassif EF, Maloney N, Conley AP, Keung EZ. Disseminated coccidioidomycosis following COVID-19 mimicking metastatic thoracic relapse of well-differentiated liposarcoma: A case report. Front Med (Lausanne) 2021;8:715939. [CrossRef]
[Pubmed]

19.
Donovan FM, Shubitz L, Powell D, Orbach M, Frelinger J, Galgiani JN. Early events in coccidioidomycosis. Clin Microbiol Rev 2019;33(1):e00112–19. [CrossRef]
[Pubmed]

20.
Fierer J, Burton DW, Haghighi P, Deftos LJ. Hypercalcemia in disseminated coccidioidomycosis: Expression of parathyroid hormone-related peptide is characteristic of granulomatous inflammation. Clin Infect Dis 2012;55(7):e61–6. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Sidak Kochar - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ahmadreza Ghasemiesfe - Conception of the work, Design of the work, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Priti Soin - Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementNone
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2026 Sidak Kochar et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.